Case History
Poster presented at the 2nd International Symposium on Dietary Therapies for Epilepsy and other Neurological Disorders, Edinburgh, Oct 2010
View pdf: EdinburghposterFinal
Refractory epilepsy controlled by an acidosis sparing, euketonemic ketogenic (ASEK) diet: a case history.
A.W.C. Yuen¹, I.A. Walcutt².
Introduction
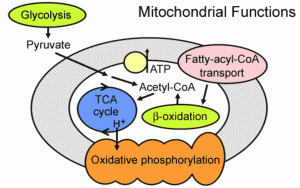 Classical ketogenic diet, the modified Atkins diet and other variants generally produce hyperketonemia (2 – 6 mM/L of beta hydroxybutyrate (BHB)) and a metabolic acidotic state. Studies undertaken so far have not been able to demonstrate a correlation between the extent of hyperketonemia and clinical efficacy. If hyperketonemia is not necessary for efficacy, might it have detrimental consequences?
Classical ketogenic diet, the modified Atkins diet and other variants generally produce hyperketonemia (2 – 6 mM/L of beta hydroxybutyrate (BHB)) and a metabolic acidotic state. Studies undertaken so far have not been able to demonstrate a correlation between the extent of hyperketonemia and clinical efficacy. If hyperketonemia is not necessary for efficacy, might it have detrimental consequences?
The human body has a number of mechanisms to maintain its pH balance, suggesting that deviations such as acidosis may have negative effects. The acidosis associated with the ketogenic diet is thought to be complicated by the formation of renal stones and osteopenia. It has also been suggested in a hypothesis that chronic acidosis may up-regulate NMDA receptors, leading to a greater likelihood of seizures occurring. This might account for the failure of the ketogenic diet in some patients. A study in 20 children on the ketogenic diet showed a significant correlation between prolonged QTc and both low serum bicarbonate and high BHB (Best TH, et al 2000). Might modifications to the classical ketogenic diet to avoid acidosis and limit the rise in ketones lead to fewer adverse effects and improve efficacy rates?
Case History
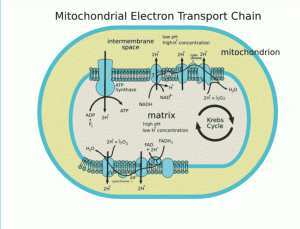 RA is a 41 yr. old female with subcortical band heterotopia. She had autism and seizures with multiple atypical seizure types which started when she was 9 months old. She was first hospitalized for status epilepticus (SE) at age 13. Her seizures remained refractory on various AED combinations. She had 8 further admissions because of SE, the last SE and hospitalization occurring at age 33. RA started ketogenic dietary treatment at age 27. Her dietary management has evolved over a period of 14 years. Her ketogenic diet has been modified for improved nutritional support, reduction of metabolic acidosis, avoidance of neurotoxins, and protection of mitochondrial and peroxisomal beta-oxidation pathways. Since age 33 years RA has been without overt episode of seizure of either a clearly defined complex partial or secondary generalized nature. EEG at age 35 was essentially normal. Primary treatment consists of phenytoin, maintained at maximum therapeutic level, high dose levetiracetam, and ketogenic-based diet (80 to 90% calories from fat). RA is now in excellent health, and shows cognitive and social rehabilitation indicative of recovery from autistic dysfunction. The tables provide a general description of RA’s acidosis-sparing, euketonemic ketogenic (ASEK) diet (Table 1), the factors that lead to reduction of acidosis (Table 2), the factors that might contribute to euketonemia (Table 3), RA’s physical examination and biochemistry results (Table 4), other management principles (Table 5), and a proposal on the biochemical effects of ASEK (Table 6).
RA is a 41 yr. old female with subcortical band heterotopia. She had autism and seizures with multiple atypical seizure types which started when she was 9 months old. She was first hospitalized for status epilepticus (SE) at age 13. Her seizures remained refractory on various AED combinations. She had 8 further admissions because of SE, the last SE and hospitalization occurring at age 33. RA started ketogenic dietary treatment at age 27. Her dietary management has evolved over a period of 14 years. Her ketogenic diet has been modified for improved nutritional support, reduction of metabolic acidosis, avoidance of neurotoxins, and protection of mitochondrial and peroxisomal beta-oxidation pathways. Since age 33 years RA has been without overt episode of seizure of either a clearly defined complex partial or secondary generalized nature. EEG at age 35 was essentially normal. Primary treatment consists of phenytoin, maintained at maximum therapeutic level, high dose levetiracetam, and ketogenic-based diet (80 to 90% calories from fat). RA is now in excellent health, and shows cognitive and social rehabilitation indicative of recovery from autistic dysfunction. The tables provide a general description of RA’s acidosis-sparing, euketonemic ketogenic (ASEK) diet (Table 1), the factors that lead to reduction of acidosis (Table 2), the factors that might contribute to euketonemia (Table 3), RA’s physical examination and biochemistry results (Table 4), other management principles (Table 5), and a proposal on the biochemical effects of ASEK (Table 6).
Table 1: General Description of ASEK
| 1. Eucaloric satisfaction of energy needs from nutrient-dense, isocaloric meals. |
| 2. Principal (80% to 90%) caloric intake from fatty acids similar in proportion to those of human milk. |
| 3. Protein restriction to approximately 1 g/kg/day with one egg, one meat, and one fish/seafood meal per day. |
| 4. Carbohydrate restriction and specificity from a list of allowed vegetables, nuts, and seeds. |
| 5. Avoidance of non-nutritive food additives, artificial sweeteners, caffeine, and alcohol. |
| 6. Sufficiency of micro nutrients for electrolyte balance, mitochondrial enzymes and cofactors. |
| 7. Meals are constructed to promote balance of oxidants and anti-oxidants. |
| 8. Adequate hydration. |
Table 2: Acidosis Sparing Factors of ASEK
| 1. Limitation of circulating acidic ketone bodies (see Table 3): acetoacetate, pKa 3.6; beta-hydroxybutyrate, pKa 4.7; acetone, pKa 20. |
| 2. Avoidance of excess amino acids. |
| 3. Avoidance of acid products of peroxidation from excess polyunsaturated and highly unsaturated fatty acids (PUFAs and HUFAs). |
| 4. High intake of alkaline-ash producing green vegetables and citrate. |
Table 3: Factors That Might Limit Hyperketonemia
| 1. Efficient beta-oxidation of fatty acids in mitochondria depends on adequate carnitine, optimal levels of enzymes and cofactors, and avoidance of inhibitors; and may be adversely affected by lowered pH. |
| 2. Efficient beta-oxidation leads to lower blood fatty acids, and reduces ketone formation in the liver. |
| 3. Restricted intake of ketogenic medium chain triglycerides. |
| 4. Efficient utilization of ketones in brain depends on low normal glucose levels, and optimal mitochondrial function. |
Table 4: RA Physical Exam and Biochemistry
| PHYSICAL EXAM | ||
| Weight | 56.6 kg | |
| Height | 159 cm | |
| Blood pressure | 120/81 mm/Hg | |
| Pulse | 83 beats/min | |
| Neurological exam | Unremarkable, except for inappropriate behaviours | |
| BIOCHEMISTRY | Normal range | |
| Sodium | 136 mEq/L | 135 – 145 |
| Potassium | 4.1 mEq/L | 3.5 – 5.2 |
| Chloride | 103 mEq/L | 95 – 109 |
| CO2 Content | 25 mEq/L | 22 – 31 |
| Glucose | 77 mg/dL (4.3mmol/L) | 70 – 99 |
| Creatinine | 0.7 mg/dL (61umol/L) | 0.6 – 1.1 |
| Calcium | 9.7 mg/dL (2.4mmol/L) | 8.6 – 10.2 |
| Lipids | ||
| HDL | 82 mg/dL (2.1mmol/L) | >59 low risk |
| LDL | 156 mg/dL (4.0mmol/L) | 130 – 159 borderline |
| Cholesterol | 250 mg/dL (6.5mmol/L) | >239 high |
| Triglycerides | 59 mg/dL (0.7mm0l/L) | <150 normal |
| CHO/HDL ratio | 3.0 | <5 desirable |
| Carnitine, free | 37 umol/L | 25 – 60 |
| Beta-hydroxybutyrate | 3.3 mg/dL (0.3mmol/L) | 0 - 3.0 |
| Ketones (not beta-hydroxybutyrate) | Negative | Negative |
Table 5: Other Important Management Factors
| 1. Hormonal regulation. |
| 2. Avoidance of constipation by supplementation with magnesium citrate to bowel tolerance. |
| 3. Management of sleep, stress and exercise. |
Table 6: Proposed Biochemical Effects of ASEK
| 1. Optimization of ATP production. |
| 2. High normal beta-hydroxybutyrate. |
| 3. Normal CO2 content/anion gap/bicarbonate concentration. |
| 4. Normal carnitine level. |
| 5. Low/normal fasting glucose level. |
| 6. Low fasting triglycerides. |
| 7. High HDL/LDL ratio. |
Conclusion
This case history suggests the potential of modification to the classical ketogenic diet, avoiding acidosis and limiting ketonemia, to improve markedly seizure control and quality of life in people with refractory epilepsy. We are aware of a female aged 10 yrs. and male aged 15 yrs. with Lennox Gastaut syndrome, and a female aged 15 yrs. with subcortical band heterotopia, who have shown marked improvement in seizure control following ASEK principles. Clearly, these anecdotal reports require further clinical experience to verify if, indeed, these principles lead to fewer adverse effects and greater efficacy.
MEDICAL REFERENCE LIST
(1) Morris RC, Jr., Schmidlin O, Frassetto LA, Sebastian A. Relationship and interaction between sodium and potassium. J Am Coll Nutr 2006 Jun;25(3 Suppl):262S-70S.
(2) Frassetto LA, Todd KM, Morris RC, Jr., Sebastian A. Estimation of net endogenous noncarbonic acid production in humans from diet potassium and protein contents. Am J Clin Nutr 1998 Sep;68(3):576-83.
(3) Ballaban-Gil K, Callahan C, O’Dell C, Pappo M, Moshe S, Shinnar S. Complications of the ketogenic diet. Epilepsia 1998 Jul;39(7):744-8.
(4) Hamed SA, Abdella MM. The risk of asymptomatic hyperammonemia in children with idiopathic epilepsy treated with valproate: relationship to blood carnitine status. Epilepsy Res 2009 Sep;86(1):32-41.
(5) Finsterer J, Segall L. Drugs interfering with mitochondrial disorders. Drug Chem Toxicol 2010 Apr;33(2):138-51.
(6) Herzog AG, Klein P, Ransil BJ. Three patterns of catamenial epilepsy. Epilepsia 1997 Oct;38(10):1082-8.
(7) Reddy DS, Rogawski MA. Neurosteroid replacement therapy for catamenial epilepsy. Neurotherapeutics 2009 Apr;6(2):392-401.
¹Dept of Clinical and Experimental Epilepsy, UCL Institute of Neurology, London, UK, and the National Society for Epilepsy, Chalfont St Peter, UK. email hidden; JavaScript is required
²Eugene, Oregon, USA. email hidden; JavaScript is required